This is a SEO version of Microsoft Word - Journal of Physics.doc. Click here to view full version
« Previous Page Table of Contents Next Page »Phosphate glass constitutes a class of optical glasses composed of metaphosphates of various metals. Instead of the SiO
4 tetrahedra observed in silicate glasses, the building block for this glass former is Phosphorus pentoxide (P
2 O
5 ), which crystallizes in at least four different forms. The most familiar polymorph (see figure) comprises molecules of P
4 O
10 .
Phosphate glasses can be advantageous over silica glasses for optical fibers with a high concentration of doping rare earth ions. A mix of fluoride glass and phosphate glass is fluorophosphate glass.
Chalcogenides
The chalcogens—the elements in group 16 of the periodic table—particularly sulfur (S), selenium (Se) and tellurium (Te)—react with more electropositive elements, such as silver, to form chalcogenides. These are extremely versatile compounds, in that they can be crystalline or amorphous, metallic or semiconducting, and conductors of ions or electrons.
Process

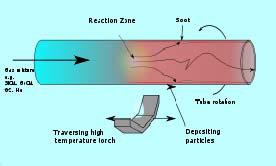
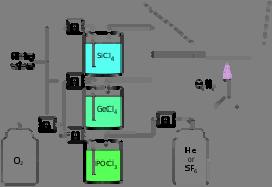

Illustration of the modified chemical vapor deposition (inside) process
Standard optical fibers are made by first constructing a large-diameter preform , with a carefully controlled refractive index profile, and then pulling the preform to form the long, thin optical fiber. The preform is commonly made by three chemical vapor deposition methods: inside vapor deposition , outside vapor deposition , and vapor axial deposition .
With inside vapor deposition , the preform starts as a hollow glass tube approximately 40 centimeters (16 in) long, which is placed horizontally and rotated slowly on a lathe. Gases such as silicon tetrachloride (SiCl
4 ) or germanium tetrachloride (GeCl
4 ) are injected with oxygen in the end of the tube. The gases are then heated by means of an external hydrogen burner, bringing the temperature of the gas up to 1900 K (1600 °C, 3000 °F), where the tetrachlorides react with oxygen to produce silica or germania (germanium dioxide) particles. When the reaction conditions are chosen to allow this reaction to occur in the gas phase throughout the tube volume, in contrast to earlier techniques where the reaction occurred only on the glass surface, this technique is called modified chemical vapor deposition .
The oxide particles then agglomerate to form large particle chains, which subsequently deposit on the walls of the tube as soot. The deposition is due to the large difference in temperature between the gas core and the wall causing the gas to push the particles outwards (this is known as thermophoresis). The torch is then traversed up and down the length of the tube to deposit the material evenly. After the torch has reached the end of the tube, it is then brought back to the beginning of the tube and the deposited particles are then melted to form a solid layer. This process is repeated until a sufficient amount of material has been deposited. For each layer the composition can be modified by varying the gas composition, resulting in precise control of the finished fiber's optical properties.
In outside vapor deposition or vapor axial deposition, the glass is formed by flame hydrolysis , a reaction in which silicon tetrachloride and germanium tetrachloride are oxidized by reaction with water (H
2 O) in an oxyhydrogen flame. In outside vapor
This is a SEO version of Microsoft Word - Journal of Physics.doc. Click here to view full version
« Previous Page Table of Contents Next Page »