This is a SEO version of Microsoft Word - Journal of Physics.doc. Click here to view full version
« Previous Page Table of Contents Next Page »simple cleaving (breaking) of the ends of the fiber can provide nicely flat surfaces with acceptable optical quality. Silica is also relatively chemically inert. In particular, it is not hygroscopic (does not absorb water).
Silica glass can be doped with various materials. One purpose of doping is to raise the refractive index (e.g. with Germanium dioxide (GeO
2 ) or Aluminium oxide (Al
2 O
3 )) or to lower it (e.g. with fluorine or Boron trioxide (B
2 O
3 )). Doping is also possible with laser-active ions (for example, rare earth-doped fibers) in order to obtain active fibers to be used, for example, in fiber amplifiers or laser applications. Both the fiber core and cladding are typically doped, so that the entire assembly (core and cladding) is effectively the same compound (e.g. an aluminosilicate, germanosilicate, phosphosilicate or borosilicate glass).
Particularly for active fibers, pure silica is usually not a very suitable host glass, because it exhibits a low solubility for rare earth ions. This can lead to quenching effects due to clustering of dopant ions. Aluminosilicates are much more effective in this respect.
Silica fiber also exhibits a high threshold for optical damage. This property ensures a low tendency for laser-induced breakdown. This is important for fiber amplifiers when utilized for the amplification of short pulses.
Because of these properties silica fibers are the material of choice in many optical applications, such as communications (except for very short distances with plastic optical fiber), fiber lasers, fiber amplifiers, and fiber-optic sensors. The large efforts which have been put forth in the development of various types of silica fibers have further increased the performance of such fibers over other materials.
Fluorides
Fluoride glass is a class of non-oxide optical quality glasses composed of fluorides of various metals. Because of their low viscosity, it is very difficult to completely avoid crystallization while processing it through the glass transition (or drawing the fiber from the
melt). Thus, although heavy metal fluoride glasses (HMFG) exhibit very low optical attenuation, they are not only difficult to manufacture, but are quite fragile, and have poor resistance to moisture and other environmental attacks. Their best attribute is that they lack the absorption band associated with the hydroxyl (OH) group (3200– 3600 cm −1 ), which is present in nearly all oxide-based glasses.
An example of a heavy metal fluoride glass is the ZBLAN glass group, composed of zirconium, barium, lanthanum, aluminium, and sodium fluorides. Their main technological application is as optical waveguides in both planar and fiber form. They are advantageous especially in the mid-infrared (2000–5000 nm) range.
HMFGs were initially slated for optical fiber applications, because the intrinsic losses of a mid-IR fiber could in principle be lower than those of silica fibers, which are transparent only up to about 2 μm. However, such low losses were never realized in practice, and the fragility and high cost of fluoride fibers made them less than ideal as primary candidates. Later, the utility of fluoride fibers for various other applications was discovered. These include mid-IR spectroscopy, fiber optic sensors, thermometry, and imaging. Also, fluoride fibers can be used to for guided lightwave transmission in media such as YAG (yttria-alumina garnet) lasers at 2.9 μm, as required for medical applications (e.g. ophthalmology and dentistry).
Phosphates
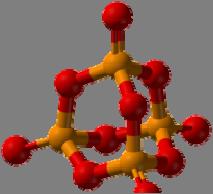






The P

4 O
10 cagelike structure—the basic building block for phosphate glass.
This is a SEO version of Microsoft Word - Journal of Physics.doc. Click here to view full version
« Previous Page Table of Contents Next Page »